DICOM family
presentation
Our DICOM family and expertise in medical imaging is at the service of your healthcare institutions and of your medical devices. The DICOM family features b<>com [DICOM-RTV Converter], a solution for synchronized real-time medical video and metadata over IP, and b<>com [Rest DICOM Library], a proxy for web-based medical image downloads. Finally, regarding the Ségur du Numérique en Santé on access to radiological examinations from the Shared Medical Record, b<>com *DICOM coaching* services allow manufacturers to benefit from b<>com's experience in understanding the specifications and development of the DRIMbox, and carry out interoperability tests.
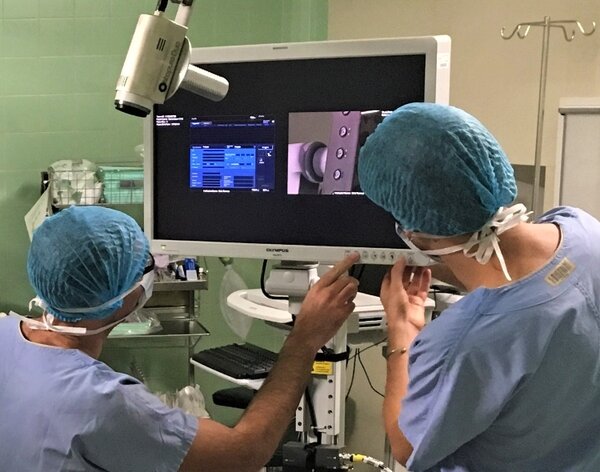
b<>com [DICOM-RTV Converter]
With the increasing number of procedures using video and live medical imaging equipment for therapeutic and diagnosis purposes, it is essential to provide the best-in-class assistance to the surgical team with smarter video and navigation systems. To ease the interoperability of such equipments inside the operating room (OR), b<>com has developed a solution that can be easily integrated into your product design.
b<>com [DICOM-RTV Converter] is a pure software module that allows the transport of uncompressed medical videos in real-time together with their associated metadata, in a synchronized way and in compliance with the DICOM-RTV standard.
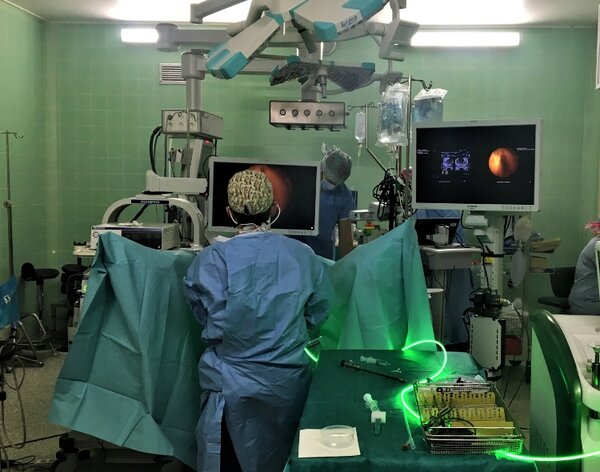
benefits
- A future proof plug and play solution.
- Guarantee of conformance with the DICOM-RTV standard.
- For OR Integrators: interoperability, integration costs reduction.
- For Video equipment providers: DICOM-RTV standard conformance, easier metadata management.
- For OR applications providers: easier video and data management while reducing the development costs.
- For Surgeons: better surgical gesture assistance thanks to synchronized multiple video and data streams.
features
- Video synchronization in real-time
- Metadata management in real-time
- Very low latency by design
- High level API to configure and operate transfers
b<>com [Rest DICOM Library]
Image sharing, at the heart of the clinical path, is rapidly increasing across hospital departments, clinics and practices with needs for remote access anywhere. The b<>com [Rest DICOM Library] is a software stack designed for Medical Imaging web uploads in accordance with the DICOMweb™ standard (STOW, WADO, QIDO). It helps strengthen collaboration between physicians, medical imaging application providers and researchers. Physicians can obtain tele-expertise from experts. Medical Imaging application providers can get easily exams to provide personalized solutions. Research organizations can obtain de-identified studies for clinical trials.
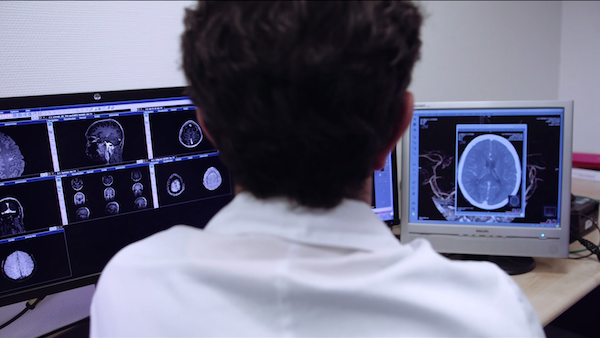
benefits
- Easy to deploy: Only one DICOM proxy to install on the server side; Zero footprint on client side
- Conform to standards: DICOM and http(s) standards
- Ready for integration: Testing results, implementation examples including GUI, documentation and support are included in the package
- Interoperability with any DICOM servers
features
- File selection with drag & drop
- Automatic parsing for grouping by study
- On-the-fly compression, JPEG-LS
- Automatic resume upload useful for large studies
- De-identification and re-identification with asymmetric key encryption

What if medical videos were automatically synchronized in real time, would that make your life easier?
certifications


